Is Nitrogen More Electronegative Than Oxygen
However this makes an oxygen group a poorer electron donating group than the corresponding nitrogen group. Water molecules have one oxygen atom that is more electronegative than the two hydrogen atoms bound to it.

Chemguide Answers Electronegativity
Postby Vasudev Tadimeti 3B Thu Dec 11 2014 146 am.

. 119 rows Values for electronegativity run from 0 to 4. Electronegativity is used to predict whether a bond between atoms will be ionic or covalent. Oxygen has 8 protons in the nucleus while nitrogen only has 7.
Answer 1 of 9. Oxygen as the more electronegative element holds more tightly to its lone pair than the nitrogen. B polar covalent bonding.
Electronegativity of oxygen is greater than nitrogen why The oxygen atom is more electronegative it is better than hydrogen at attracting electrons because i. A bonding pair of electrons will experience more attraction from the oxygens nucleus that from nitrogens thus the electronegativity of oxygen is greater. Fluoride is a stronger field ligand than the other halogens but ammonia is a stronger field ligand than water.
While it is also derived from the Zeff its a slightly different use of the term. Nitrogen is less electronegative than oxygen it is in better position to accommodate the positive charge on the proton. This table is a.
This explains why Oxygen is. A bonding pair will experience more attraction from the oxygens nucleus than from nitrogens and so the electronegativity of oxygen is greater. Is N or as more electronegative.
But oxygen has 8 protons in the nucleus whereas nitrogen only has 7. Both A and R are true and R is the correct explanation of A. I dont exactly have figures to tell u but oxygen is more electronegative than nitrogenThink logically electronegatIviy is e the average of ionisation enthalpy and election gain enthalpy In case I of nitrogen ionisation enthalpy is highly positive and electron gain enthalpy is.
A bonding pair will experience more attraction from the oxygens nucleus than from nitrogens and so the electronegativity of oxygen is greater. Since oxygen is more electronegative than hydrogen the two bonds that are formed will be polar covalent which means that a partial negative charge will be on the more electronegative atom oxygen and two partial positive. A non-polar covalent bonding.
As a result which type of bonding holds water molecules together. In fact oxygen is second only to fluorine in this respect. Electronegativity means that the atom is less willing to give up the electrons.
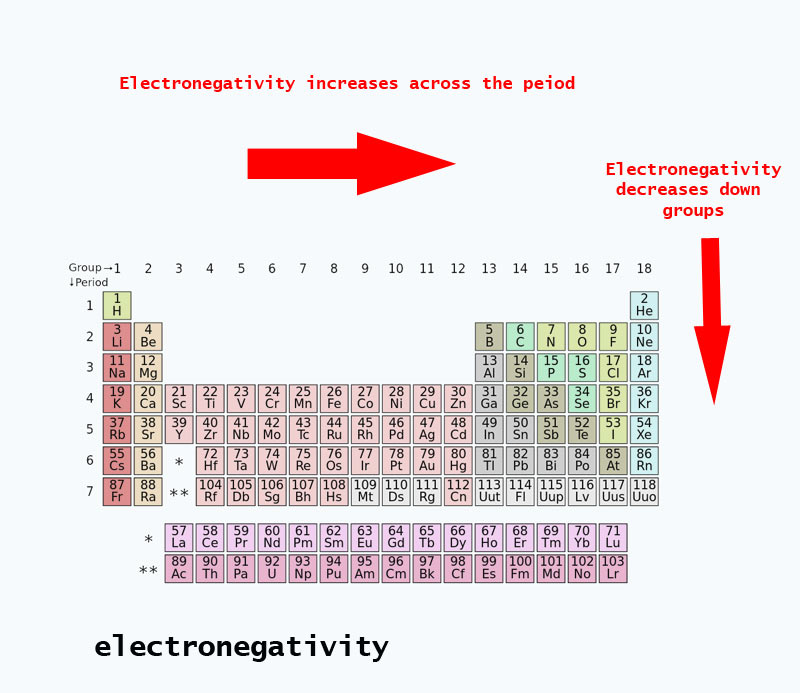
Why is oxygen more electronegative than nitrogen. And thus across a Period from RIGHT to LEFT as we face the Table fluorine is more electronegative than oxygen which is more electronegative than nitrogen which is more electronegative than carbon which is more electronegative than boron. Oxygen is higher in atomic number.
A Oxygen is more electronegative than nitrogen fluorine is more electronegative than the other halogens. Oxygen is more electronegative and thus has a higher ionization energy than nitrogen. Oxygen is more electronegative than nitrogen but nitrogen has higher Ionisation Energy than oxygen.
No oxygen is more electronegative than nitrogen. The nitrogen lone pair therefore is more likely to break away and form a new bond to a proton it is in other words more basic. Amines are more basic than ethers and esters.
Oxygen as the more electronegative element holds more tightly to its lone pair than the nitrogen. As you go to the right on the Periodic Table. So sulphur is less electronegative than oxygen.
Nitrogen is more willing to give up donate its electrons because it is less electronegative. Similar with electron affinity describing the amount of energy released to the surroundings when an electron is attached to a gaseous neutral atom. The bonds between nitrogen and oxygen are covalent bonds made from sharing electron pairs.
And thus across a Period from RIGHT to LEFT as we face the Table fluorine is more electronegative than oxygen which is more electronegative than nitrogen which is more electronegative than carbon which is more electronegative than boron. Oxygen has 8 protons in the nucleus whereas nitrogen only has 7. Which elements are more electronegative than oxygen.
Is o2 more polar than h2. And thus across a Period from RIGHT to LEFT as we face the Table fluorine is more electronegative than oxygen which is more electronegative than nitrogen which is more electronegative than carbon which is more electronegative than boron. But oxygen has 8 protons in the nucleus whereas nitrogen only has 7.
A bonding pair will experience more attraction from the oxygens nucleus than from nitrogens and so the electronegativity of oxygen is greater. Oxygen is more electronegative than nitrogen but nitrogen has higher ionization energy than oxygen Why. Fluorine is more electronegative than the other halogens.
Electronegativity for Oxygen and Nitrogen. Fluoride is a stronger field ligand than the other halides but ammonia is a stronger field ligand than water. Oxygen is more electronegative than nitrogen.
It can also be used to predict if the resulting molecule will be polar or nonpolar. The nitrogen lone pair therefore is more likely to break away and form a new bond to a proton it is in other words more basic.

Chemguide Questions Electronegativity
Why Does Carbon Gain Electrons In Co When Oxygen Is More Electronegative Than C Quora

Why First Ionization Enthalpy Of Nitrogen Is More Than Oxygen
Why Does Oxygen Nitrogen And Chlorine Have High Electronegativity Quora

Why Is Nitrogen More Stable Than Oxygen Quora
Which Element Is More Electronegative Hydrogen Or Sulfur Quora
Why Does Nitrogen Have Less Electron Affinity Than Oxygen Quora

Electronegativity And Oxidation Number Introduction To Chemistry
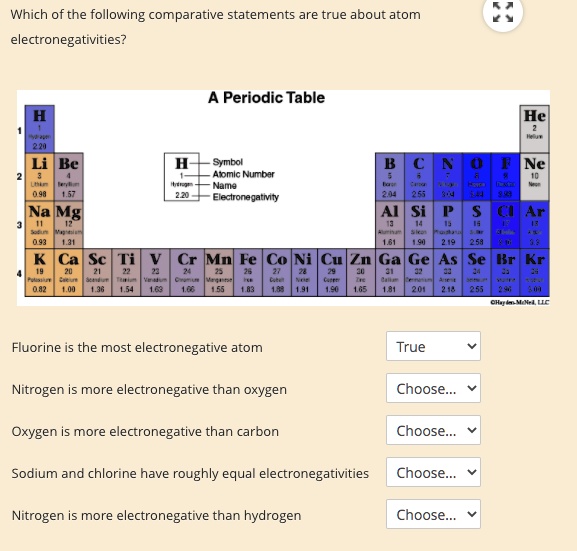
Solved Which Of The Following Lom Parative Statements Are True About Atom Electronegativities Periodic Table He Be Symbol Apmic Numbar Nama Electronegatmty Net Na Mg Ar Derar Ca Sc Mn Co Ni Cu
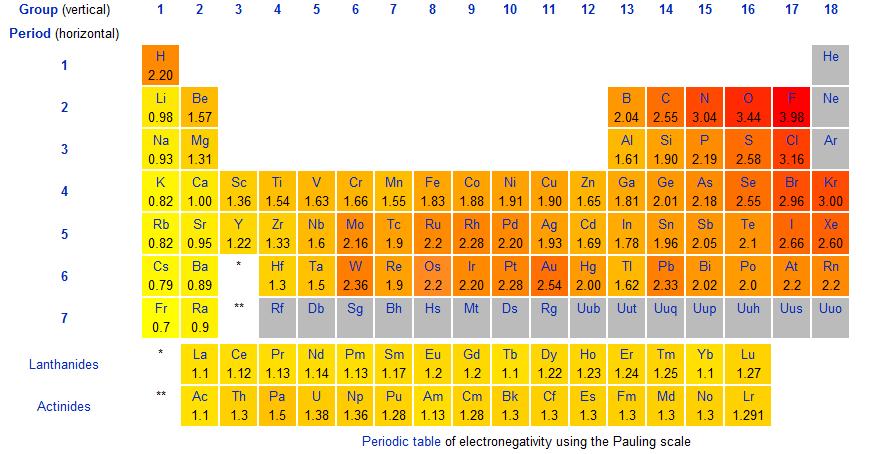
What Is Electronegativity Definition Chart And Trends

Water And Polarity Chapter Ppt Video Online Download
Which Is More Electronegative Bromine Or Iodine Quora
Why Does Oxygen Nitrogen And Chlorine Have High Electronegativity Quora
Why Does Oxygen Nitrogen And Chlorine Have High Electronegativity Quora
Which Is More Electronegative Oxygen Or Nitrogen Quora
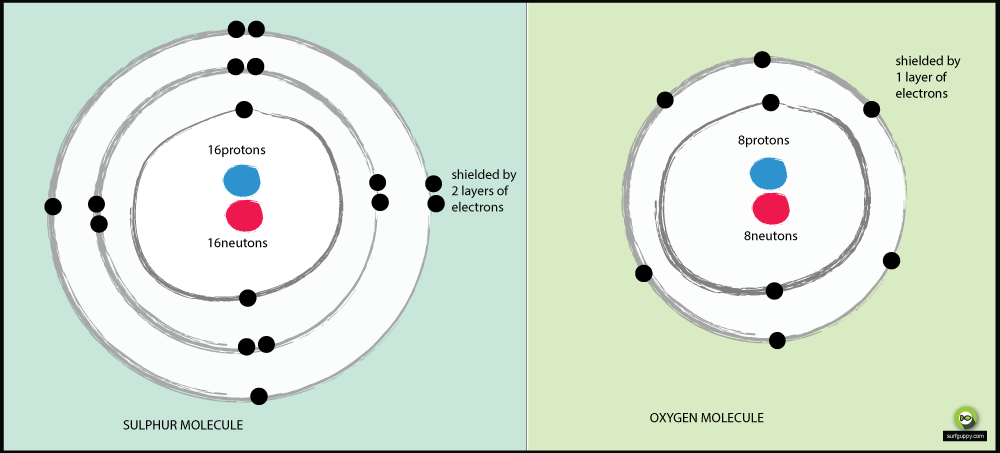
Comments
Post a Comment